Closed-Cell vs. Open-Cell Sponges
Apr 23rd 2026
In a radiology department that meticulously tracks radiation dose, inspects protective garments annually, and follows strict hand hygiene protocols, there’s often one item that slips through the infection control conversation: the positioning sponge. These foam blocks, wedges, and cylinders are used dozens of times a day to support patients during imaging, and in many facilities, they’re quietly harboring exactly the kind of contamination that infection control programs are designed to prevent.
Understanding the Two Foam Types
Not all foam is created equal, and the difference between the two types used in radiology positioning couldn’t be more consequential for infection control.
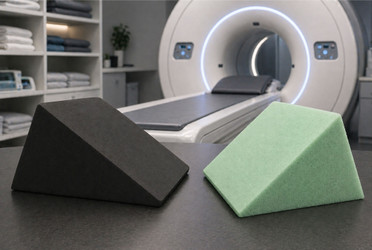
Open-cell foam (non-coated) is the familiar spongy material most people associate with cushioning. Its structure is porous and made up of interconnected cells that allow air and liquid to pass freely through the material. Think of a kitchen sponge. Open-cell foam absorbs blood, barium, Betadine, body fluids, and cleaning solutions. Once absorbed, those fluids are nearly impossible to fully remove. The foam becomes a reservoir for bacteria, mold, and fungal growth, with contamination building up invisibly over weeks and months of use.
Closed-cell foam (crosslink polyethylene) has a fundamentally different structure. Each cell is a sealed, self-contained unit. There are no interconnecting channels for liquid to penetrate. The result is a material that is completely non-porous and cannot absorb fluids even when the surface is punctured or torn. This is the critical distinction: an open-cell sponge with a tear is a contamination highway, while a closed-cell sponge with a tear still provides a barrier because the internal cell structure remains sealed.
The Infection Control Implications
In a radiology suite, positioning sponges contact patient skin, hospital gowns, wound dressings, and occasionally open surgical sites. They are used on every table, in every room, across every shift. A single sponge might contact a dozen different patients in a day.
If that sponge is open-cell foam, it has been absorbing trace amounts of body fluid from every patient interaction. Standard surface wiping, even with hospital-grade disinfectant, cannot reach the interior of a porous material. The surface may appear clean while the interior remains contaminated. This is not a theoretical concern; it’s a well-documented failure mode in infection control.
Closed-cell foam eliminates this pathway entirely. Because the material cannot absorb fluid, contamination remains on the surface where it can be effectively reached by disinfectant wipes. Techno-Aide’s closed-cell sponges are compatible with standard medical-grade disinfectants including PDI Sani-cloth AF3 alcohol-free germicidal wipes which are the same wipes already in use throughout most radiology departments.
Beyond Infection Control: Durability and Imaging Performance
Closed-cell crosslink polyethylene foam has significantly higher tensile and tear strength than open-cell alternatives. In practical terms, this means closed-cell sponges last dramatically longer before needing replacement. They resist the compression set that causes open-cell sponges to lose their shape over time, maintaining consistent patient positioning support throughout their service life.
Both foam types used in radiology are radiolucent, meaning they allow X-rays to pass through without significant artifact. However, open-cell sponges that have absorbed barium or contrast media can produce unexpected radiopaque artifacts on subsequent exams which is another contamination-related issue that closed-cell foam avoids entirely.
Closed-cell foam is also antimicrobial, antibacterial, antifungal, and ozone-resistant. It will not support the growth of mold, mildew, or bacteria on its surface or interior claim that open-cell foam simply cannot make.
The Total Cost Equation
Open-cell foam sponges are typically far less expensive per unit. This leads many departments to treat them as disposable or semi-disposable items, replacing them when they become visibly stained or misshapen. But “less expensive per unit” doesn’t mean less expensive over time.
When you factor in the replacement frequency (closed-cell sponges last three to five times longer), the infection risk (one HAI costs a hospital tens of thousands of dollars), and the imaging artifact potential, closed-cell foam is often the more economical choice on a total cost-of-ownership basis.
A Look at Techno-Aide’s Sponge Lineup
Techno-Aide offers positioning sponges across the full spectrum of clinical needs and price points, including Closed Cell sponges for maximum infection control, Vinyl-Coated sponges that combine a foam core with a heavy-gauge, fluid-resistant vinyl cover, Coated sponges (Stealth Cote) featuring a proprietary medical-grade cleanable coating, and Non-Coated sponges for applications where infection control requirements are lower.
In our next post, we’ll take a closer look at the Stealth Core and Stealth Cote technologies that make Techno-Aide’s antimicrobial sponge lines stand out from standard closed-cell products.